Outside of getting excited about playoff hockey and snowboard trips out west, I also find myself enthusiastic about studies/efforts in cancer biology that push the boundaries to further characterize pre-clinical cancer models in a clinical way. The study presented in this summary does exactly that, and wouldn’t be possible without collaboration across multiple groups.
The use of fiducial markers in image-guided radiotherapy (IGRT) is a common practice to help clinicians accurately target tumors located within the abdominal area. Tumors located in areas such as the gastrointestinal tract, pancreas, and liver can be difficult to identify with CT alone. Furthermore, small locations like the prostate can be displaced when comparing the daily CT scan to the planning CT scan. Because of this, fiducial markers are used to identify the actual location of the lesion. This technique allows for more accurate radiation delivery while minimizing normal tissue toxicity.
The transrectal implantation of fiducial markers is a common method when treating prostate tumors with IGRT. An effective method for accurate treatment, fiducial markers can often cause pain during and symptoms of discomfort following implantation. Furthermore, the use of metal fiducial markers can cause streaking artifacts when acquiring a cone beam-CT. Because of this, a novel non-toxic liquid fiducial marker called BioXmark (Nanovi, Kongens Lyngby, Denmark) has recently been developed for both clinical and pre-clinical use. BioXmark is ready to inject and is composed of ethanol as a solvent, which diffuses after injection leaving a semi-solid hydrophobic gel. Originally developed for clinical use, the characterization of BioXmark in small animal cancer models is minimal.
Despite continuous clinical and pre-clinical research efforts, pancreatic tumors remain the 4th leading cause of death in the U.S with a 5-year survival rate of less than 5% (Jemal et al. Cancer Statistics. 2010). Because of this, research focused on novel treatment methods are necessary to improve clinical outcome. The capability of the SARRP to combine high-resolution cone beam-CT imaging with targeted radiation delivery has enabled researchers to perform studies not otherwise possible. With the emergence of novel fiducial markers like BioXmark, combination studies with SARRP allow for even more accurate modeling and further complete the translational research model. In an effort to further show the feasibility of use and effectiveness in small animal cancer models. A group led by Dr. Stephanie E. Combs at the Technical University of Munich further investigated BioXmark in a murine orthotopic pancreatic tumor model.
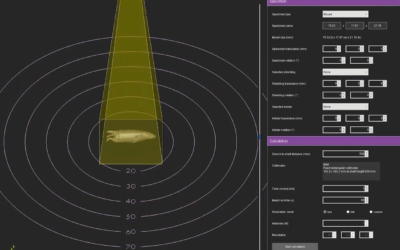
The purpose of this study was not only to show the functionality of BioXmark in an orthotopic pancreatic murine tumor model but also to identify optimal volumes of injection and its stability long term. It was observed that small volumes of BioXmark (10µl) could be detected by CBCT (Figure 1). However, volumes exceeding 50µl resulted in hardening artifacts and is suggested not to exceed 50µl in preclinical mouse models. With volumes between 10-25µl being the most optimal for injection and image quality. BioXmark was also observed to be stable 4 months after injection, showing the benefit of use in long-term studies (Figure 2). BioXmark was also found to be beneficial in registering the planning CBCT with the daily CBCT when performing fractionated studies.
In a previous blog post, I summarized a study out of University of Pennsylvania by Verginadis et al. titled “A novel mouse model to study image-guided, radiation-induced injury and preclinical screening of radioprotectors.” In this study, researchers developed a novel method by surgically implanting a radiopaque marker for guidance in targeting a small portion of the intestinal tract via CBCT. An effective method for image guidance, however, induced experimental variables resulting in the death of up to 10% of the mice that underwent surgery. The authors of the present study reference this observation in the full manuscript and expand on the fact that no animals were lost due to side effects from injection of BioXmark. Therefore, it was concluded that BioXmark offers a safer and non-toxic option compared to surgical implantation fiducial markers as well as iodinated contrast enhancement agents.
When SARRP was originally developed at Johns Hopkins University, the idea was to push the envelope of preclinical radiation based studies. Being translational as possible in small animal cancer modeling while keeping pace with clinical advancements is imperative to move forward effectively in the fight against cancer. Continuous development and characterization of agents like BioXmark with SARRP reflect the continued effort of Xstrahl, the researchers using SARRP, and collaborators like Nanovi who develop BioXmark. I’m looking forward to what’s next on the horizon, and encourage the SARRP community to dig deep in their pre-clinical investigations.
This study establishes the first orthotopic pancreatic tumor mouse model combining SARRP and a fiducial marker. The full article can be accessed at https://www.ncbi.nlm.nih.gov/pubmed/28808749
BioXmark is a proprietary agent manufactured by Nanovi http://nanovi.com/bioxmark/
Dobiasch S, Kampfer S, Burkhardt R, Schilling D, Schmid TE, Wilkens JJ, Combs SE. BioXmark for high-precision radiotherapy in an orthotopic pancreatic tumor mouse model: Experiences with a liquid fiducial marker. Strahlenther Onkol. 2017
This Xstrahl In Action was adapted from a article found on a National Library of Medicine website.